NVIC’s 2023 Annual Report on U.S. State Vaccine Legislation
By NVIC Advocacy Staff
Published November 02, 2023
NVIC's Annual State Vaccine Legislative Report
Read more
.png?lang=en-US&ext=.png)
ACIP Recommends RSV Vaccination for Pregnant Women
By Theresa Wrangham
Published September 28, 2023
RSV Vaccine Recommended for Pregnant Women
Read more
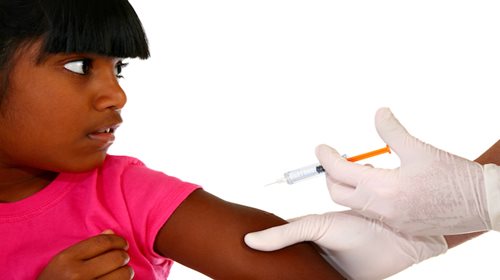
School-Based Health Centers & Minor Consent to Vaccination
By Theresa Wrangham
Published August 24, 2023
Learn about School Based Health Centers offering vaccination services to students and federal requirements that parents receive vaccine information before vaccination.
Read more

Back to School - Vaccine Education, Requirements & Exemptions
By Theresa Wrangham
Published August 17, 2023
Learn about school vaccine requirements, exemptions and national policy being developed to teach children about vaccines in the classroom.
Read more
.png?lang=en-US&ext=.png)

