Read and report vaccine reactions, harassment and failures.
Chickenpox (Varicella) Disease & Vaccine Information
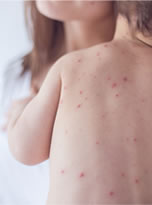
Below are brief introductions to chickenpox (varicella) and the chickenpox (varicella) vaccine with links to more information. Scroll down for a list of QUICK FACTS that provide a summary overview of key facts for the disease and the vaccine.
Chickenpox: The Disease
Chickenpox (varicella) is an illness caused by the varicella zoster virus, a DNA virus that is part of the herpes virus family and associated with shingles. The virus first presents as a chickenpox infection; however, if the virus reactivates, it will present as a shingles infection. Chickenpox is transmitted through direct contact with chickenpox blisters, by inhaling particles from the chickenpox blisters, and possibly from contact with respiratory secretions infected with the virus. Symptoms of chickenpox generally begin between 10 and 21 days following exposure to the virus and the illness typically lasts between 5 and 10 days. The varicella virus is found only in humans and outbreaks generally occur between March and May in the United States. Learn more about Chickenpox…
Chickenpox Vaccine
There are two live virus vaccines for chickenpox licensed for use in the U.S. by the FDA: Varivax, a live varicella (chickenpox) virus vaccine manufactured by Merck; and ProQuad (MMRV), a live virus vaccine containing measles, mumps, rubella and varicella (chickenpox), also manufactured by Merck. There is a higher risk of seizure following MMRV vaccination if this vaccine is administered as the first dose, when compared to the administration of separate doses of MMRII and varicella vaccines. The CDC recommends children receive a first dose of chickenpox vaccine between 12 and 15 months and a second dose between 4 and 6 years. The CDC also recommends two doses of chickenpox vaccine be administered 4 to 8 weeks apart to all school-aged students, post-secondary aged students, and all healthy adults who lack immunity to chickenpox. Learn more about Chickenpox vaccine…
Chickenpox Quick Facts
Chickenpox
-
Chickenpox (Varicella) is a highly-contagious infection that is generally mild in most children;
-
Chickenpox is transmitted through direct contact with, or by inhaling particles from chickenpox blisters. It may also be spread through respiratory secretions of a person infected with the virus;
-
Recovery from chickenpox confers long lasting natural immunity and immunocompetent individuals rarely experience a second attack. Re-exposure to chickenpox has been found to boost immunity and reduce the risk of shingles infection in older children and adults. Continue reading quick facts…
Chickenpox Vaccine
-
Mild side effects, such as redness, rash, or pain at the injection site, as well as fever, have been reported following chickenpox vaccination. More serious side effects of chickenpox vaccine include meningitis, pneumonia, seizures, full body rash, allergic reaction, and death. Mild side effects following MMRV vaccination include rash, redness, or pain at the injection site, fever and swelling of the glands in the neck or cheeks. More serious side effects of MMRV vaccine may include loss of hearing, meningitis, pneumonia, full body rash, seizure, coma, brain damage, severe allergic reaction, and death;
-
Chickenpox vaccine is reported to be between 80 and 85 percent effective at preventing chickenpox and more than 95 percent effective at preventing severe illness. The widespread use of chickenpox vaccine in the U.S. has substantially increased the rate of shingles infections in adults, as a natural boost of immunity from exposure to chickenpox in the environment is no longer occurring; Continue reading quick facts…
NVIC encourages you to become fully informed about chickenpox and the chickenpox vaccine by reading all sections in the table of contents, which contain many links and resources such as the manufacturer product information inserts, and to speak with one or more trusted health care professionals before making a vaccination decision for yourself or your child. This information is for educational purposes only and is not intended as medical advice.



